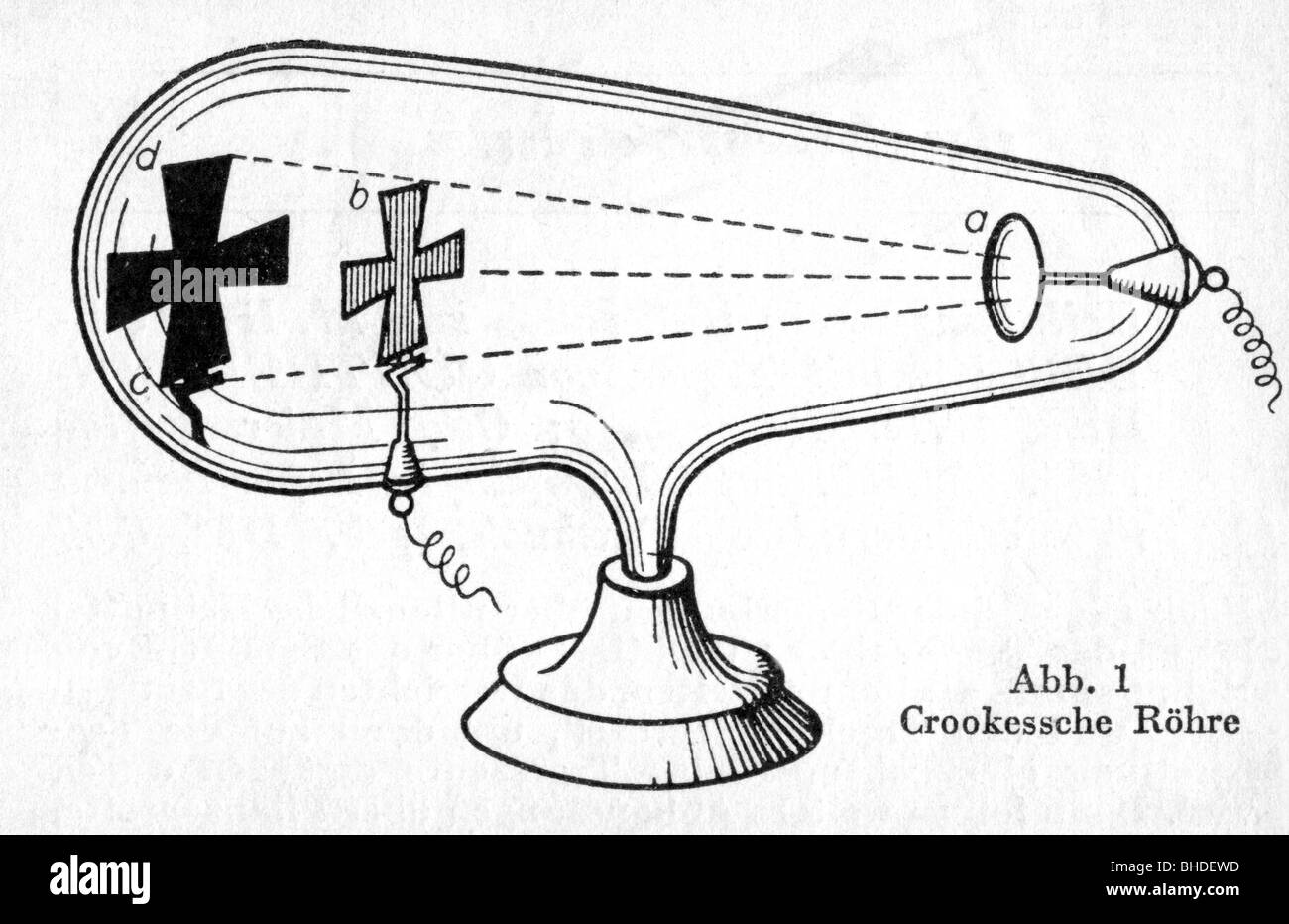
The force applied on an electron due to electric field is given by F =qE. Why do electrons deflect?ĭeflection of electron due to electric field Cathode rays are formed when electrons emitted from one electrode and travel to another when a voltage is applied in a vacuum. Thomson determined that charged particles much lighter than atoms, particles that we now call electrons made up cathode rays. 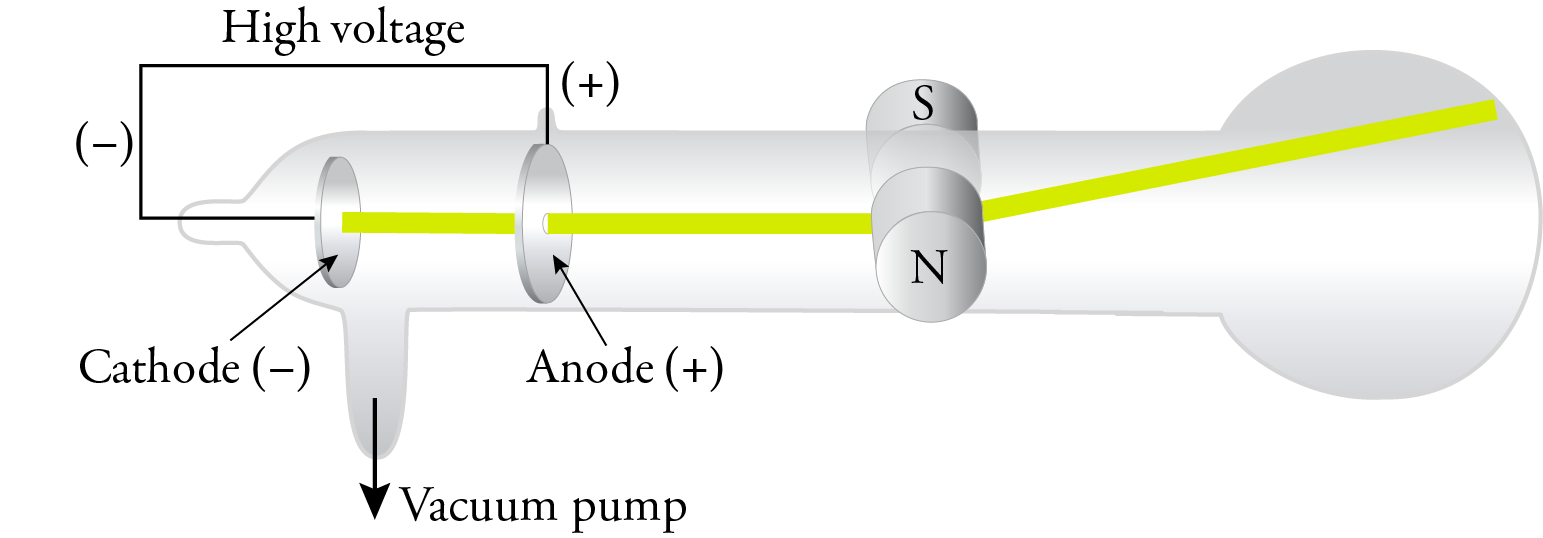
Thomson, in full Sir Joseph John Thomson, (born December 18, 1856, Cheetham Hill, near Manchester, England-died August 30, 1940, Cambridge, Cambridgeshire), English physicist who helped revolutionize the knowledge of atomic structure by his discovery of the electron (1897). This experiment also provided an estimate of the ratio of the charge to the mass of these particles. Thomson)īy balancing the effect of a magnetic field on a cathode-ray beam with an electric field, Thomson was able to show that cathode “rays” are actually composed of particles. Thomson discover the charge to mass ratio? He formulated the first atomic theory since the “death of chemistry” that occurred during the prior 2000 years. Who is responsible for the 200 year death of chemistry?ĭeath to the field of Chemistry for 2000 years! In the early 1800s, the English Chemist John Dalton performed a number of experiments that eventually led to the acceptance of the idea of atoms. Cathode rays were determined to be composed of negatively charged particles that were smaller than the smallest atom. Prior to the experiment, it was not known that atoms were composed of further particles. Thomson’s cathode ray tube experiment discovered the subatomic particle the electron. Thomson’s cathode ray tube experiment show about atoms? Who made the plum pudding model of an atom? What did J.J. What major contribution did Thomson make to the Atomic Theory? Discovered that atoms can be split into smaller particles (protons and electrons). Thomson’s plum pudding model of the atom had negatively-charged electrons embedded within a positively-charged “soup.” What did Thomson contribute to atomic quizlet? Thomson’s experiments with cathode ray tubes showed that all atoms contain tiny negatively charged subatomic particles or electrons. What did the cathode ray tube experiment demonstrate? The electron charge-to-mass ratio was measured by accelerating the electrons through a voltage Vx towards a positively charged plate. He determined that the negatively charged particles (electrons) were much lighter than the positively charged particles. 
In addition, he also studied positively charged particles in neon gas. He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube. He observed that cathode rays were deflected by a magnetic field in the same manner as a wire carrying an electric current, which was known to be negatively charged. In order to determine if the cathode ray consisted of charged particles, Thomson used magnets and charged plates to deflect the cathode ray. What did Thomson use to deflect the cathode rays? Thomson realized that the accepted model of an atom did not account for the particles charged negatively or positively. He had shown that the cathode rays were charged negatively. Thomson invented the electron by playing with a tube that was Crookes, or cathode ray. How did the cathode ray tube help discovered the electron? How did Thomson use the cathode ray tube in his research quizlet?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
